After a great conversation with Erick Turner yesterday I thought I would make an attempt at a rapid review. Erick wrote the seminal paper Selective publication of antidepressant trials and its influence on apparent efficacy. In this paper Erick used FDA regulatory documents to highlight significant discrepancies between reviews based on published journal articles and regulator data. Nothing I’ve seen makes me think that reviews based purely on published articles – for drug efficacy estimates – can deliver anything other than ballpark estimates. Worse than that, the dangers of this reliance on published articles is hidden from consumers (doctors, nurses, patients, policy makers etc) – this is verging on immoral.
The above rant aside I thought I’d have a go at a rapid review based on FDA data. I have been collecting FDA and EMA regulatory data for inclusion in the Trip Database and picked the first 2015 review I came across and that happened to be on Brexpiprazole and was examined for two uses – schizophrenia and as an adjunct for major depressive disorder. To keep things simple I decided to concentrate of it’s use in schizophrenia. So, here goes…:
Brexpiprazole for schizophrenia
Background: Brexpiprazole (proposed proprietary name “Rexulti”) is a new molecular entity atypical antipsychotic co-developed by Otsuka Pharmaceutical Co, Ltd (Otsuka) and H. Lundbeck A/S (Lundbeck). It has partial agonist activity at serotonergic 5-HT1A and at dopaminergic D2 receptors, antagonist activities at serotonergic 5-HT2A, and antagonist activity at noradrenergic α1/2 receptors.
Primary efficacy endpoint: Mean reduction in PANSS total score at week 6.
Number of trials: 2. Study 230 = 657 patients, Study 231 = 623 patients.
Results:
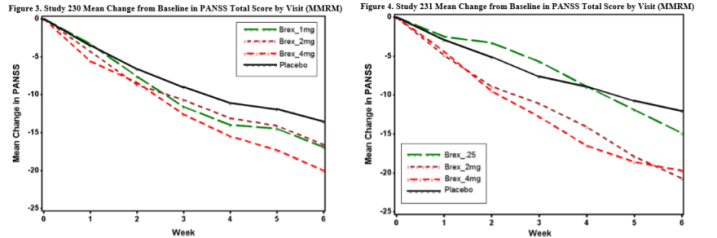
Adverse events: In short-term, controlled trials, the percentage of subjects reporting >1 treatment-emergent adverse events (TEAEs) was similar in the brexpiprazole 2-4 mg/day, all brexpiprazole, and placebo groups (59.2%, 60.6%, and 61.1% respectively). Events reported by >5% of subjects the all brexpiprazole group included headache, akathisia, insomnia, and agitation. Only akathisia was reported by a higher percentage of subjects in the all brexpiprazole group than in the placebo group, and it did not reach the “most common” threshold of >5% and >2x placebo.
References:
- FDA Medical Review
- FDA Statistical Review
- Study 230: A multicenter, randomized, double-blind, controlled phase 3 trial of fixed-dose brexpiprazole for the treatment of adults with acute schizophrenia. Schizophr Res. 2015 May;164(1-3):127-35
- Study 231: Efficacy and Safety of Brexpiprazole for the Treatment of Acute Schizophrenia: A 6-Week Randomized, Double-Blind, Placebo-Controlled Trial. Am J Psychiatry. 2015 Sep 1;172(9):870-80
So, some comments from me:
- It took around 20-30 minutes to produce
- The EMA have yet to publish anything on brexpiprazole
- There is a systematic review: Brexpiprazole for schizophrenia and as adjunct for major depressive disorder: a systematic review of the efficacy and safety profile for this newly approved antipsychotic – what is the number needed to treat, number needed to harm and likelihood to be helped or harmed? Int J Clin Pract. 2015 Sep;69(9):978-97. The conclusions are effectively the same.
- The two trials in Brexpiprazole have both been published, which is perhaps unsurprising as they are both positive. It’ll be interesting to see what the results are for another review were trials are unpublished
An important point is that I’m not publishing the above as some sort of perfect solution. It’s there as a very quick test to start using regulatory data to create meaningful reviews. As I repeat the process things will change etc. But please comment if you have anything constructive to say!
3 thoughts on “Example rapid review: Brexpiprazole for schizophrenia”