The second rapid review produced using regulatory data, the first being Brexpiprazole for schizophrenia.
Empagliflozin in Type 2 Diabetes
Background: Empagliflozin is a reversible, potent and selective competitive inhibitor of sodium-glucose co-transporter 2 (SGLT2). SGLT2 is highly expressed in the kidney (proximal tubules) where it is responsible for 90% of glucose reabsorption from the glomerular filtrate back into the circulation. Inhibition of SGLT2 reduces renal reabsorption of glucose resulting in reduction of glucose in blood.
Overview of clinical program: The clinical development program for empagliflozin consisted of 48 clinical trials including 30 Phase I trials, 5 dose-finding Phase II trials, and 13 Phase IIb/III trials. Empagliflozin was evaluated as monotherapy and in combination with metformin, glimepiride, pioglitazone, insulin and dipeptidyl-peptidase 4 (DPP-4) inhibitors (sitagliptin). Clinical trials included 13767 patients; 6808 patients were treated with empagliflozin for at least 24 weeks, 4415 for at least 52 weeks and 1486 for at least 76 weeks. All cardiovascular events were independently adjudicated and evaluated in a prospective meta-analysis.
Primary endpoint: Change from baseline in glycosylated hemoglobin (HbA1c).
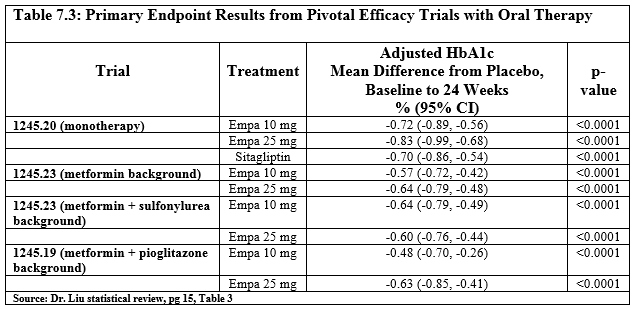
Efficacy: Empagliflozin is effective in reducing glycosylated hemoglobin (HbA1c) when used as monotherapy and as add-on to other antidiabetic regimens (including metformin, metformin + sulfonylureas, pioglitazone, and basal insulin). The adjusted mean difference in HbA1c from placebo ranged between -0.48% and -0.73% for empagliflozin 10 mg and between -0.59% and -0.84% for empagliflozin 25 mg after 24 weeks of treatment; the decrease in HbA1c was sustained for at least 52 weeks. In addition, empagliflozin 25 mg demonstrated non-inferiority to glimepiride (1 mg to 4 mg). Observed changes in fasting plasma glucose and in postprandial glucose support the changes observed in HbA1c. There was reduced efficacy of empagliflozin in patients > 75 years old and in patients with severe renal impairment. Empagliflozin also demonstrated significant and clinically meaningful reductions in HbA1c when compared to placebo in subpopulations of diabetic patients who also had hypertension, renal impairment, or increased cardiovascular risk and in patients who were already receiving insulin therapy. Furthermore, when compared to placebo, treatment with empagliflozin also resulted in reduction in body weight and systolic blood pressure.

Adverse events:

Cardiovascular safety: The primary safety endpoint of this meta-analysis was MACE+, a composite endpoint comprising CV death, non-fatal myocardial infarction (MI), non-fatal stroke, or hospitalization for unstable angina. The FDA document was heavily redacted but states “Based on the current data, there does not appear to be an increased risk of CV events…”
The EMA’s EPAR reports “The primary endpoint of this analysis was the composite 4-point MACE (major adverse cardiovascular events) endpoint, which consisted of cardiovascular death (including fatal stroke and fatal myocardial infarction), non-fatal myocardial infarction, non-fatal stroke, and hospitalisation due to unstable angina.” They further reported “For the primary endpoint based on the treated set, the incidence was 1.98% for the placebo group (26 patients with event) and 0.92% in the all empagliflozin group (22 patients with event), with an incidence rate of 22.2 events (placebo) vs 9.8 events (all empagliflozin) per 1000 years at risk. The hazard ratio (HR) [95% CI] based on Cox regression of all empagliflozin vs placebo was 0.48 [0.27, 0.85].”
References
Comments
- The first review took twenty minutes, this took closer to an hour.
- These reviews are a snapshot early on in the evidence cycle. The Trip Medical Director was surprised to not see much in the way of mortality data. This was because the initial trials, those used to get approval, had the primary outcome of reduced HbA1c.
- New evidence is added all the time and mortality safety was the primary outcome of a subsequent NEJM paper, reported in the BMJ Blogs. Which reports “Although powered as a non-inferiority study, empaglifozin demonstrated statistical superiority as add-on diabetic therapy, significantly reducing the incidence of the primary end-point (10.5% vs. 12.1%, HR 0.86; 95% CI, 0.74 to 0.99; P=0.04 for superiority), including a significant reduction in the risk of death (5.7% and 8.3%, respectively; 32% relative risk reduction).“
- So, after an initial review of the regulatory data it’s not particularly difficult (and something Trip is exploring) to have an active monitoring of new trials/reviews and adding data as and when it becomes available.
- In traditional systematic reviews the whole SR needs to be started again including the full search, screening of all trials, assessment of bias etc. That’s a lot of work. By scanning – on a prospective basis – for new data means updating can take place very quickly. To put that in to context, Cochrane approaching this would have taken two years to do the initial review of papers. Then, as subsequent articles were published, another two years. Using regulatory data is so much quicker and then a more pragmatic updating system means you have up to date reviews with 1% of the effort. I really fail to see what 99% of the effort buys you!
- The hour was quite quick and I do think a much better job could be done with a bit longer – but I wanted to see how quick it could be done. Imagine what you could do with a whole day!
2 thoughts on “Example rapid review: Empagliflozin in Type 2 Diabetes”